Infection Prevention Orientation Manual
Section 12: Antibiotic Stewardship
Karen Burk, RPh
October 2014
Download a printable PDF Version of this section.
Objectives
At the completion of this section the Infection Preventionist (IP) will:
- Learn how to generate, interpret and distribute the antibiogram for the facility
- Determine who is responsible for reviewing antimicrobial use in the facility
- Assist in development of an antimicrobial stewardship program for the facility
- Understand how to interpret the culture and sensitivity report
- Develop a list of contacts for the facility
Number of hours
- Antibiogram Section: 4 Hours
- Antimicrobial Stewardship Section: 4 Hours
- Culture and Sensitivity Report Section: 2 Hours
Suggested Readings
- Core Elements of Hospital Antibiotic Stewardship Programs. Atlanta, GA: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention; 2014. Available at: www.cdc.gov/getsmart/healthcare/implementation/core-elements.html .
- Hindler JF, Stelling J. Analysis and Presentation of Cumulative Antibiograms: A New Consensus Guideline from the Clinical and Laboratory Standards Institute. Clinical Infectious Diseases. 2007; 44(15):867-873. Available at: oxfordjournals.org/content/44/6/867.full.pdf+html
- Clinical and Laboratory Standards Institute (CLSI) M39-A: Guidelines For Reporting A Cumulative Antibiogram (Current Edition)
- Dellit TH, Owens RC, McGowan JE, et al. Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America Guidelines for Developing an Institutional Program to Enhance Antimicrobial Stewardship. Clinical Infectious Diseases. 2007; 44(15):159-177. Available at: idsociety.org/uploadedFiles/IDSA/Guidelines-Patient_Care/PDF_Library/Antimicrobial%20Stewardship.pdf
- Leekha S, Terrell CL, Edson RS. General principles of antimicrobial therapy. Mayo Clinic Proceedings. 2011; 86(2): 156–167. Available at: ncbi.nlm.nih.gov/pmc/articles/PMC3031442/
- Wyoming Infection Prevention Orientation Manual (WY IPOM) Laboratory Section (#10) and Microbiology Section (#11)
Overview
Antibiotic use today directly impacts effectiveness tomorrow. Antibiotics used in single patient can have a direct impact on another single patient; they are a shared resource. Antibiotic resistance is not just a problem for the person with the infection. Some resistant bacteria have the potential to spread to others, promoting antibiotic-resistant infections. It takes a long time to develop antibiotics effective in treating resistant infections. It is imperative to improve upon the use of antibiotics currently available. Three main concepts covered in this chapter are: the antibiogram, the antimicrobial stewardship program (ASP), and the bacterial culture and antibiotic sensitivity report (aka culture and sensitivity).
Key Terms
Table 1. Key terms in antimicrobial stewardship and the pharmacy.
Key Concepts
The Antibiogram
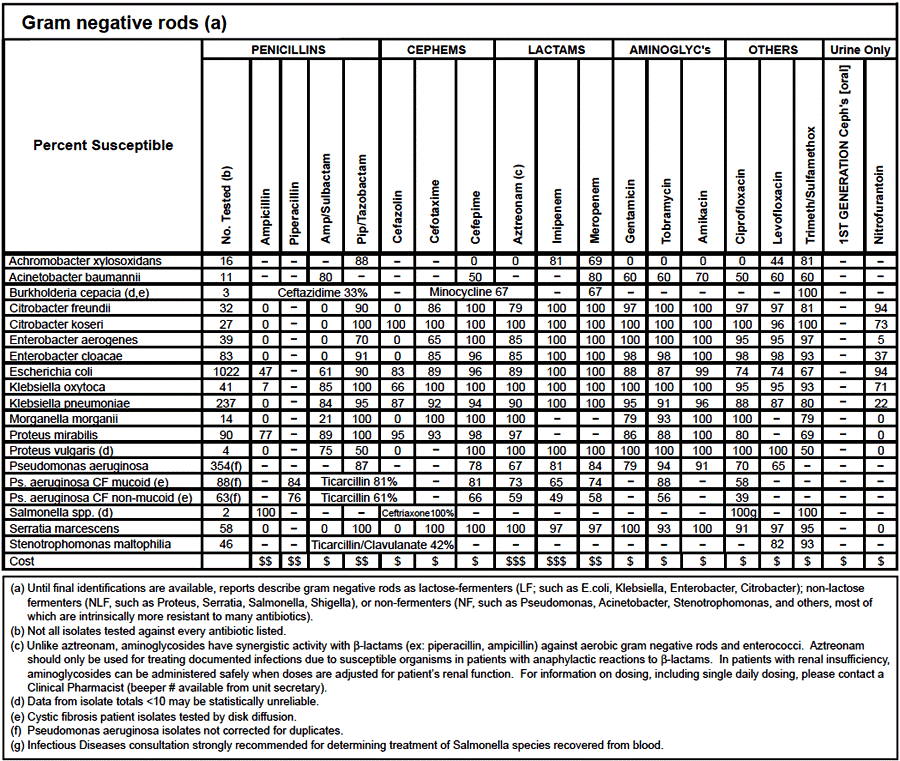
An antibiogram is the result of laboratory testing for the susceptibility of an isolated bacterial strain to antibiotics in a defined period of time, typically six to twelve months for a particular institution. Cumulative susceptibility results are organized into a summary table, also known as a cumulative antibiogram which is often simply called the antibiogram. A typical antibiogram shows the total number of bacterial isolates tested against a range of antimicrobials (Figures 1 and 2). The antibiogram includes the percentage (%) of bacterial isolates susceptible to each antimicrobial agent tested and are often reported by inpatient or outpatient status, as well as by individual wards such as intensive care unit, medical surgical unit, or long term care facility. It is usually divided by types of organisms including Gram-negative bacteria, anaerobes, and Gram-positive bacteria. Urine cultures are also often separated out to help prevent skewing the overall susceptibility results. The antibiogram may also contain additional information to help guide the healthcare provider into appropriate empiric antibiotic selection for that specific institution as well as showing relative cost of the various antimicrobials (Figures 1 and 2). Bacterial susceptibility patterns often change by location; hence, each institution will have its own unique antibiogram. It is for this reason that another institution’s antibiogram results cannot be applied at another facility unless they are in close proximity to one another and/or share the majority of patients/residents.
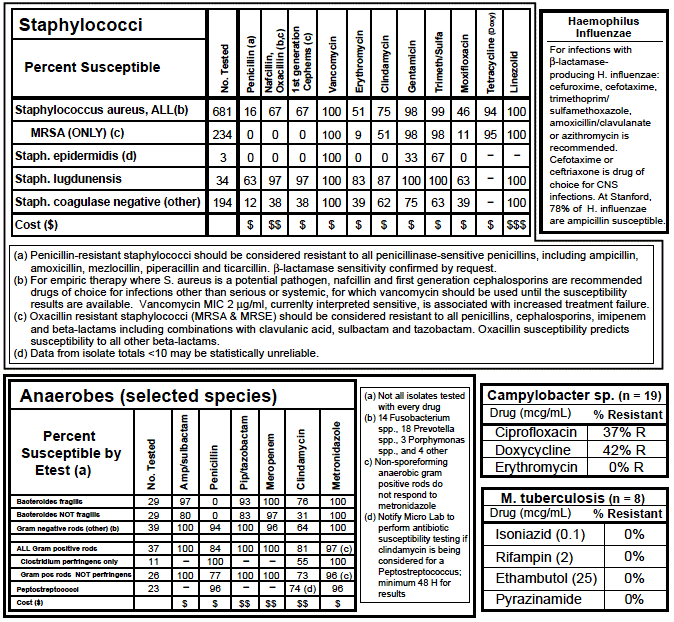
Figure 2: Sample antibiogram from Standford School of Medicine, Stanford University medical Center. Used with permission. Available at: errolozdalga.com/medicine/pages/OtherPages/stanfordAntibiogram.html. Accessed February 7, 2014.
The primary purposes of the antibiogram are:
- To guide the empiric selection of antimicrobials
- To use as an educational tool for prescribers
- To monitor antibiotic resistance trends in bacteria common among patient/resident populations and in the community
The antibiogram can be used as a reference guide by physicians, infection prevention personnel, pharmacists, microbiologists, and nurses to show resistance and susceptibility patterns to various organisms at their institution and increases the likelihood that the patient has the best chance to receive the correct antibiotic. This results in improved patient outcomes, cost savings for both the patient and the institution, and a decrease in antimicrobial resistance. Once the culture and sensitivity report is back from the laboratory, clinicians should rely on this data rather than the antibiogram for ongoing treatment.
The antibiogram can also raise awareness of resistance trends in the institution. It can identify opportunities to reduce inappropriate antibiotic use which can potentially result in treatment failures. For example, if the institution’s fluoroquinolone susceptibility rate decreases from the previous year, the institution could create guidelines and/or restrictions to aid the physician in appropriately prescribing fluoroquinolones. The judicious use of an antibiotic will improve the susceptibility rate over time, allowing it to be more effective for future patients. The percentage susceptible for a given species of organism will be impacted by several factors including culturing practices, patient population, specimen collection practices, and laboratory antimicrobial-susceptibility testing policies. Overprescribing of antibiotics and inappropriate prescribing practices may also lead to increased resistance patterns.
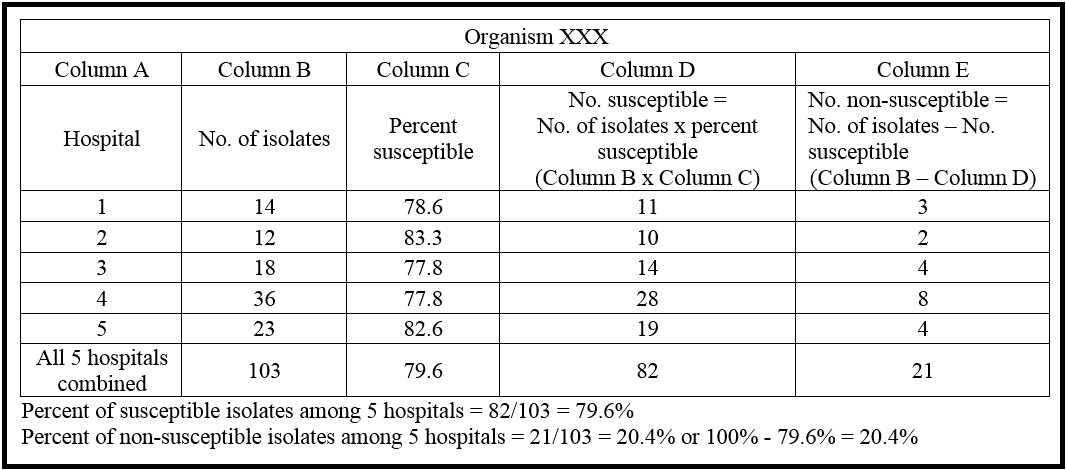
The CLSI has developed recommendations for the collection, analysis, and presentation of cumulative antimicrobial susceptibility test data for the antibiogram. These recommendations lead to better comparability among institutions and minimize the tendency to overestimate drug-resistance. The CLSI recommends a minimum of 30 isolates per species of organism for an antibiogram to be considered valid. Smaller institutions may not meet this recommendation, and in that case, data from multiple facilities can be merged to create one antibiogram. This pooling of data from neighboring institutions may be useful for providing a general guide for resistance patterns in the community. Caution must be used when reviewing pooled data, as the susceptibility profiles can vary significantly among institutions even when in close proximity particularly if there is a vast difference in type of patient population. For example, children’s hospitals may have different resistance patterns compared to long term care facilities. Certain population subgroups may be over represented and need to be excluded from the community antibiogram. Figure 3 shows an example of how aggregated data can be used from multiple facilities.
Figure 3: Example of how data from multiple antibiograms can be aggregated for an organism to estimate susceptibility or non-susceptibility in a community
The Antimicrobial Stewardship Program (ASP)
Antimicrobial stewardship is an integral part of improving patient care and minimizing resistance to antimicrobials. Antimicrobial stewardship (as defined by the Society for Healthcare Epidemiology of America [SHEA], the Infectious Diseases Society of America [IDSA], and the Pediatric Infectious Diseases Society [PIDS]) includes coordinated interventions designed to improve and measure the appropriate use of antimicrobial agents.1 This can be accomplished by promoting the selection of the optimal antimicrobial drug regimen, which includes dosing, duration of therapy, and route of administration. The major objectives of an ASP are to achieve best clinical outcomes related to antimicrobial use while minimizing toxicity and other adverse events.1 As such, an ASP will limit the selective pressure on bacterial populations that drive the emergence of antimicrobial-resistant strains. Antimicrobial stewardship may also reduce excessive costs attributable to suboptimal antimicrobial use.
There are two core approaches to antimicrobial stewardship: “front-end” (a.k.a. pre-prescription) and “back-end” (a.k.a. post-prescription). The front-end pre-prescription approach uses restrictive prescriptive authority by restricting certain antimicrobials and requiring prior authorization either before their use or within a certain timeframe. The back-end, or post-prescription approach uses prospective review and feedback by reviewing current antibiotic orders, then makes recommendations to continue, adjust, change, or discontinue the antimicrobial therapy based on laboratory results.
There are four reasons for implementing an ASP: 1) Antibiotic resistance is a significant and progressively worsening problem at healthcare facilities globally. This fact, combined with the lack of new antimicrobial agents in the drug development pipeline, indicates that judicious antimicrobial management is necessary to preserve the effectiveness of antibiotics currently available. 2) Adverse outcomes result from the inappropriate choice, dose, formulation, or duration of antibiotics. Adverse outcomes include increased cost of antibiotics, antibiotic resistance, increased morbidity, mortality, and length of stay. Increased costs associated with antibiotic use include drug expenditures and extended lengths of stay. 3) Stabilizing antibiotic resistance requires a multipronged approach including formulary restrictions, education, and review of antimicrobial prescribing with close surveillance of antibiotic utilization and resistance patterns. 4) Inappropriate antimicrobial use is strongly associated with the emergence of resistant pathogens. An ASP is effective in the emergence and transmission of antimicrobial-resistant organisms.1, 2 The appropriate use of antimicrobials is an essential part of patient safety, improving patient care and shortening hospital stays.
The following strategies contribute to an effective ASP.
Strategy #1. Creation of a multidisciplinary antimicrobial stewardship team. Ideally, a team is directed by an infectious disease physician and a clinical pharmacist. The team will have a clinical microbiologist, an information system specialist, an IP, and a hospital epidemiologist. In rural healthcare facilities, committee members might include a physician (preferably with some infectious disease training), a pharmacist, a microbiologist, and an IP. A nurse, or employees, (preferably from pharmacy, lab, and infection prevention), interested in ASP will suffice. It would be very helpful to find a physician champion as well.
Exercise #1
Exercise #1: Identify members are also members of the ASP. Recommended members:
- Infection Preventionist
- Microbiologist
- Physician
- Pharmacist
- RN – Med./Surg.
- RN – LTCC
- RN – Clinic
- RN – ICU
Strategy #2. IP-based strategies. Infection preventionist based strategies for antimicrobial stewardship often include developing a process to measure and monitor antimicrobial use, including obtaining reports from the laboratory, comparing results to the patient’s antimicrobial and determining appropriate use. The IP, the microbiologist, and the pharmacist work together to develop an antibiogram based on antimicrobials available. The IP distributes the facility-specific antibiogram to the ASP, healthcare providers, and important stakeholders.
Strategy #3. Pharmacist-based strategies. Pharmacists employ protocols and prescribing guidelines that help optimize the patient antibiotic regimens. Protocols and guidelines are approved through the pharmacy and therapeutics committee and forwarded to the medical staff for review and approval. Local quality patient care based on regional population and resistance patterns should supersede national guidelines. One example protocol to implement would be a pharmacist driven parenteral to oral conversion protocol (when the patient’s condition allows) in the selection of antimicrobials or antifungals with excellent bioavailability. Examples of such antimicrobials could include the fluoroquinolones, linezolid, metronidazole, clindamycin, trimethoprim-sulfamethoxazole, fluconazole, and voriconazole. Changing from parenteral to oral therapy can result in reduced length of stay, health care costs, and potential complications due to intravenous access.3
Pharmacists are an excellent resource to help address dose optimization. Many pharmacist driven aminoglycoside and vancomycin protocols utilize pharmacist’s expertise which results in more rapid attainment of the correct dose, thus maximizing the chance of cure, and minimizing the risk of drug toxicity.4
Pharmacists play a key role in ensuring that antimicrobial orders have correct dose, correct duration, and correct indication. They can address inappropriate duplicate antimicrobial therapy. Pharmacists and the IP can both monitor for antibiotic duration longer than 7 days. Duration of antibiotic therapy in the hospital setting is often longer than necessary. Longer durations of antimicrobial therapy tend to promote super-infections with organisms that are more resistant. It is imperative to take an “antibiotic timeout” to reassess the antibiotic(s) the patient is receiving. Therapy should be de-escalated whenever possible to a narrow spectrum antimicrobial in order to help prevent resistance development to the broad spectrum antimicrobials. Vancomycin and carbapenem duration greater than 3 days should be reviewed, as empiric therapy with these agents in severely ill patients is often reasonable initially but can be subsequently discontinued.
Strategy #4. Surveillance of antibiotic utilization and resistance patterns. Antibiotic utilization and resistance patterns should be monitored. The IP and pharmacist work together to compile quarterly data on antibiotics. The ASP committee (or the IP if there is no ASP committee) provides feedback to healthcare providers on a unit. A potential problem in antibiotic resistance is high rates of Clostridium difficile infections.
Strategy #5. Antibiogram based guidelines. Facilities must utilize their own current antibiograms to develop guidelines for their institution. A formulary limited to non-duplicative antibiotics with demonstrated clinical need will reduce over-prescribing of unnecessary antimicrobials. Physicians may use and recommend appropriate antimicrobials for disease states based on the current antibiogram.
Strategy #6. Laboratory based strategies. Laboratory personnel can also assist with antimicrobial stewardship. The microbiologist monitors resistance patterns and trends and notifies the IP when needed. Working with pharmacists, the laboratory personnel can help encourage healthcare providers to obtain cultures prior to administering antibiotics. Nursing staff often coordinate this effort.
Strategy #7. Information Technology (IT)-based strategies. Information technology (IT) personnel may assist in electronic surveillance system development for identifying patients receiving inappropriate antimicrobial therapy. Electronic alerts identify patients whose antibiotic therapy does not “match” the reported microbiologic susceptibilities of the patients’ organisms (“bug-drug mismatch alert”). Electronic alerts can serve as reminders to physicians and nurses to verify cultures have been performed before the medication is administered.
Strategy #8. Leadership support. Facility administrative support is essential for improving the “buy-in” of clinical staff for stewardship interventions and programs. Formal statements include stewardship-related duties in job descriptions, annual performance reviews, ensuring staff from relevant departments are given sufficient time to contribute to stewardship activities, support training and education of the entire staff, and encourage participation from facility staff.
Strategy #9. Education. Education regarding the correct use of antimicrobials should be provided to all staff members, the patients, family members, and visitors. ASP should provide national and local updates on antibiotic prescribing, antibiotic resistance, and infectious disease management.5. Communicating information on antibiotic use motivates the potential for improved prescribing.6 Providing education can be done formally and informally via posters, flyers, newsletters, and electronic communication. Reviewing de-identified cases with providers is useful. Education has been found to be most effective when paired with corresponding interventions and measurement of outcomes.1 A variety of web-based educational resources can help facilities develop education content.7, 8 The Centers for Disease Control and Prevention (CDC) has several slide shows available for download. See the Overview and Evidence to Support Stewardship section at www.cdc.gov/getsmart/healthcare/evidence.html.
Monitoring process or outcome measures allows the IP to track the success of interventions. The selection of outcome measures that are important to key groups can be very helpful, but these will need to be tailored for that particular group. For example, antibiotic costs might be an important measure for administrators but are not compelling for clinicians. Reductions in Clostridium difficile infections or adverse events are likely to be more important for clinical audiences. While resistance is an important endpoint and a topic of interest for clinicians, most experts in antibiotic stewardship agree that it is not an ideal primary endpoint, as resistance rates change very slowly and can be influenced by a variety of factors, in addition to antibiotic use.9 The CDC has developed a program called the Antibiotic Stewardship Driver Diagram and Change Package – A Framework to Reduce Inappropriate Antibiotic Utilization in Hospitals as seen in Appendix A and available at: www.cdc.gov/getsmart/healthcare/implementation.html . The CDC also has a program entitled Core Elements of Hospital Antibiotic Stewardship Programs. Please see their website: www.cdc.gov/getsmart/healthcare/implementation/core-elements.html for more information and a checklist to assess key elements and actions to ensure ASP compliance (Appendix B).
Exercise #2
The Bacterial Culture and Sensitivity Report
Culture and sensitivity (C&S) testing identifies pathogens, provides information regarding the effectiveness of antimicrobials, and takes 48-72 hours. Preliminary tests conducted within 24 hours include the Gram stain. Refer to the WY IPOM Sections #10, Laboratory and #11, Microbiology for more information. Through interpretation of C&S results, physicians confirm antibiotic selection and an alternative or additional agent. Appendix C provides a list of organisms and appropriate antimicrobial agents. It is important to note that when choosing empiric antimicrobials, consider the source of infection. For example, E. coli is a common organism found in urinary tract infections. The use of an empiric antimicrobial would cover Gram-negative bacteria. When the laboratory report is available, the narrower spectrum antibiotic should be prescribed.
Exercise #3
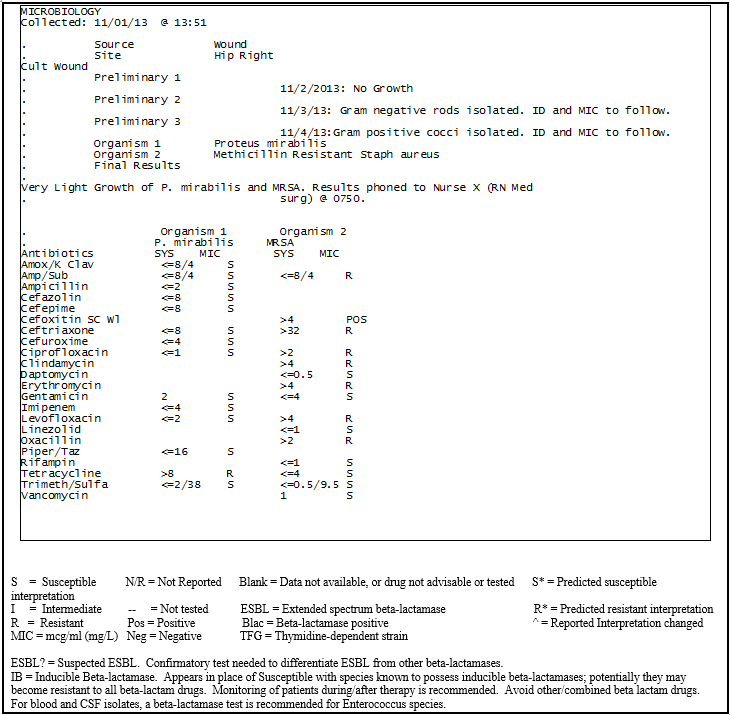
A C&S report contains the name of the organism, source of the specimen, antimicrobials used to treat that organism, the sensitivity to each agent (e.g., S = sensitive, I = intermediate, R = resistant), and if the organism is a suspected extended spectrum beta-lactamase (ESBL) producer. If there is a suspected ESBL organism, the physician will want confirmation prior to giving an antimicrobial as it may actually be resistant in vivo if it is true. If the organism is proven not to be an ESBL producer, the antimicrobial agent may be utilized. The report will specify if the organism has inducible beta-lactamase (IB) properties, indicated as (S) for susceptible next to the antimicrobial (for example ceftriaxone) but will also have “IB” listed after the (S). Some laboratories may list IB next to the MIC value of the organism. Though the clinician may initially choose this antimicrobial as it appears sensitive, it will rapidly become resistant due to the beta-lactamase properties of the organism (as will other beta-lactams). Unlike the potential ESBL producing organism, any antimicrobial listed with IB should be avoided; including other beta-lactams. Figure 4 shows an example of how an organism with potential ESBL or inducible beta-lactamase properties could be displayed.
Final C&S reports show the MIC value adjacent to the sensitivity interpretation (S, I or R). The MIC is the minimum concentration at which an antimicrobial inhibits visible growth of the organism. The report does not provide information regarding whether the organism is actually killed. Susceptibility in vitro does not uniformly predict clinical success in vivo. If the organism is resistant, this will often but not always, correlate with treatment failure. The only true measure of bacterial response to an antibiotic is the clinical response of the patient. A report of “susceptible” indicates that the isolate is likely to be inhibited by the usually achievable concentration of an antimicrobial agent when the recommended dosage is used. For this reason, MICs of different agents for a particular organism are not directly comparable.10 A common misconception when interpreting the C&S report is to assume the choice antibiotic is the one with the lowest MIC number. In reality, the MIC interpretations are specific to both the organism and the antimicrobial agent. For example, ciprofloxacin achieves serum concentrations of 1 to 4 mcg/mL and ceftriaxone achieves peak serum concentrations of 100 to 150 mcg/mL. An MIC of 4mcg/mL for either antibiotic against E. coli would be interpreted on the C&S report as resistant to ciprofloxacin and susceptible to ceftriaxone. Likewise, MICs of 1mcg/mL (susceptible) for ciprofloxacin and 2 mcg/mL (susceptible) for ceftriaxone against E. coli, do not imply that ciprofloxacin is twice as active as ceftriaxone. Instead, it indicates that concentrations achieved by giving recommended doses of both drugs are likely to be active against the organism.10 Interpretation of quantitative susceptibility tests is based on the relationship of the MIC to the achievable concentration of antibiotic in body fluids with the dosage given for a given organism. Do not assume the antimicrobial with the lowest MIC is always the best one to choose!!
Several methods are available for determining the dose of antimicrobial needed. The most common approach to antibiotic dosing is to adjust doses to obtain antibiotic plasma concentrations above the MIC for the respective pathogen throughout the dosing interval.11 Antibiotics are frequently divided into two major groups: those that exhibit time-dependent (concentration-independent) killing and minimally to moderately persistent effects and those that exhibit concentration-dependent killing and prolonged persistent effects.23 For antibiotics belonging to the first group (beta-lactam antibiotics, vancomycin, and macrolides), their effect depends on the length of time the drug is in contact with the bacteria. Effects will increase with increasing concentrations until the maximum kill rate is reached. After that point, increasing concentrations will not produce a corresponding increase in the effect. Maximum killing occurs at concentrations approximately four to five times the MIC.11 The second group of antibiotics, which include the aminoglycosides and fluoroquinolones, exhibit a different killing pattern; bacterial rates of killing increase with increasing concentrations of the antibiotic. The goal in this case is to maximize the drug concentration. Parameters currently used are those which reflect an increase in drug concentration, i.e., Cmax/MIC (the ratio between the peak concentration [Cmax] of the antimicrobial and the MIC).11 This parameter is the relationship between the maximum drug concentration reached in the patient at steady state and the MIC established for the pathogen responsible for the infection.23 A different approach to assess the antimicrobial efficacy of antibiotics is pharmacokinetic-pharmacodynamic models based on time-kill curves. Time-kill curves follow microbial killing and growth as a function of both time and antibiotic concentration.11 The advantage of these in vitro models is that they allow direct comparison of concentration profiles and provide for a much more detailed assessment of the pharmacokinetic-pharmacodynamic relationship than the simple use of MICs.11
When determining which antimicrobial to choose it is important for clinicians and laboratory personnel to be aware of the site of infection. For example, an isolate of Staphylococcus aureus could be reported as susceptible to cefazolin in vitro. However, if this particular isolate was obtained from the cerebrospinal fluid (CSF), cefazolin would not be an optimal therapeutic choice because it does not achieve therapeutic concentrations in the CSF.22 It is good practice to communicate directly with the microbiologist when antimicrobial susceptibility patterns appear unusual. Be aware of limitations in the antimicrobial susceptibility testing. Testing of relatively newer agents such as daptomycin for Gram-positive cocci, might not be routinely performed or reported but could be available on request.
In Figures 4 and 5, the sample C&S report laboratory indicates what is occurring in the sample specimen daily. As seen in Figure 5, there was no growth on day 1, day 2 shows a Gram-negative rod identified, and day 3 shows an additional organism, a Gram-positive cocci was isolated. By checking these reports, the physician can tailor the antimicrobial(s) based on the results. Once an organism is identified as a multi-drug resistant organism (in this case methicillin resistant S. aureus [MRSA]), the microbiologist should notify the nurse. The nurse should then contact the physician (if the laboratory hasn’t done so already) to review the antibiotics. The nurse should also ensure the patient is placed under the appropriate isolation precautions. If the IP is diligent in reviewing the daily C&S reports, he/she will be aware of these situations and can educate nursing staff about the required precautions.
Figure 5: An example of a culture and sensitivity report for multiple organisms isolated.
Methods
Generation of the antibiogram
The laboratory will provide the data for the antibiogram. The clinical microbiologist, physician, pharmacist, epidemiologist, or the IP might be the person to analyze and present the antimicrobial susceptibility data. Several staff members may work together to generate the antibiogram. The CLSI recommends several bacterial isolate and antimicrobial agent criteria that should be considered when generating an antibiogram. The CLSI recommends including:
- final, verified, clinical cultures from humans
- antibiotics routinely tested against the species of interest
- the first isolate of a given organism per patient per analysis period regardless of body site or susceptibility profile
- bacterial species with at least thirty isolates tested bacterial isolates that report as susceptible to the specified antibiotic when calculating percentages. Do not include isolates with intermediate sensitivity except for Streptococcus pneumoniae, viridans streptococci, and Staphylococcus aureus as per the Clinical and Laboratory Standards Institute M39-A manual.
- Calculate and list both the percentage susceptible and the percentage of isolates with intermediate susceptibility for penicillin for Streptococcus pneumoniae. In addition, calculate and list the percent susceptible for cefotaxime or ceftriaxone using both the meningitis and non-meningitis breakpoints. Also report the percentage susceptible to oral penicillin if applicable for the institution.
- For viridans streptococci, calculate and list both the percentage susceptible and the percentage of isolates with intermediate susceptibility for penicillin.
- For Staphylococcus aureus, calculate and list the percentage susceptible for all isolates, as well as for the subset of methicillin resistant Staphylococcus aureus.
The CLSI recommends excluding:
- isolates from surveillance cultures such as Methicillin-Resistant Staphylococcus aureus (MRSA) nasal swabs
- multiple isolates of the same organism from the same patient in the analysis period
Cumulative antibiograms should be prepared annually. More frequent reporting or distribution may be necessary if large numbers of isolates or noticeable changes in susceptibilities are present. Consider reporting antibiogram results every six months. A new antibiogram for the facility may be necessary if newer antimicrobial agents have been added or replaced older agents. Smaller facilities having fewer than 30 isolates may only generate an antibiogram every other year. Antimicrobial agents reported include only those routinely tested and clinically useful against the population of isolates to be analyzed. Do not include data for antibiotics that are clinically inappropriate for an organism despite in vitro susceptibility (e.g., first generation cephalosporins and Salmonella). CLSI guidelines provide a table for antibiotic vs. organism on pages 34-43 of the M100-S23 manual.
The clinical laboratory often uses a commercial data management computer system or develops their own software to analyze the facility’s cumulative susceptibility data. This software is typically integrated with the laboratory computer system to manage data collected and generated by the clinical laboratory. The laboratory computer system may interface with the facility information system. The CLSI M39-A manual recommends analysis and presentation of cumulative antimicrobial susceptibility test data for both required and desirable characteristics of the analysis database. The analysis database should include the results for all antimicrobials tested, including those agents not routinely reported. This avoids bias introduced by selective reporting practices. Data should be retained for surrogate testing but reported on the antibiogram as the percent susceptible for the agent represented by the surrogate. For example, the use of a cefoxitin disk to check for MRSA should be reported as the percent susceptible for oxacillin. Verification for the removal of duplicate data isolates will need to be performed. Line listings of susceptibility data should be compared with computer-generated reports to ensure accurate calculation. This will need to be done initially, when changes are made to the software or the MIC/disk diffusion interpretive criteria, and for select organisms where multiple cultures are often performed.12 To verify all duplicate isolates have been removed, provide the isolate sensitivity report list by patient name. The person compiling the antibiogram can scan for duplicates and exclude them.
The facility may place a dash or black box in the column of an antibiotic next to the organism to show which antibiotics were not tested due to intrinsic resistance of the organism, the antibiotic not included on the panel used by the laboratory, or the facility does not have the medication on formulary. This helps the physician select only those antibiotics appropriate and available for the organism. Figure 2 on page 7 also shows an example of intrinsic resistance or antimicrobials not tested.
Interpretation of the Antibiogram
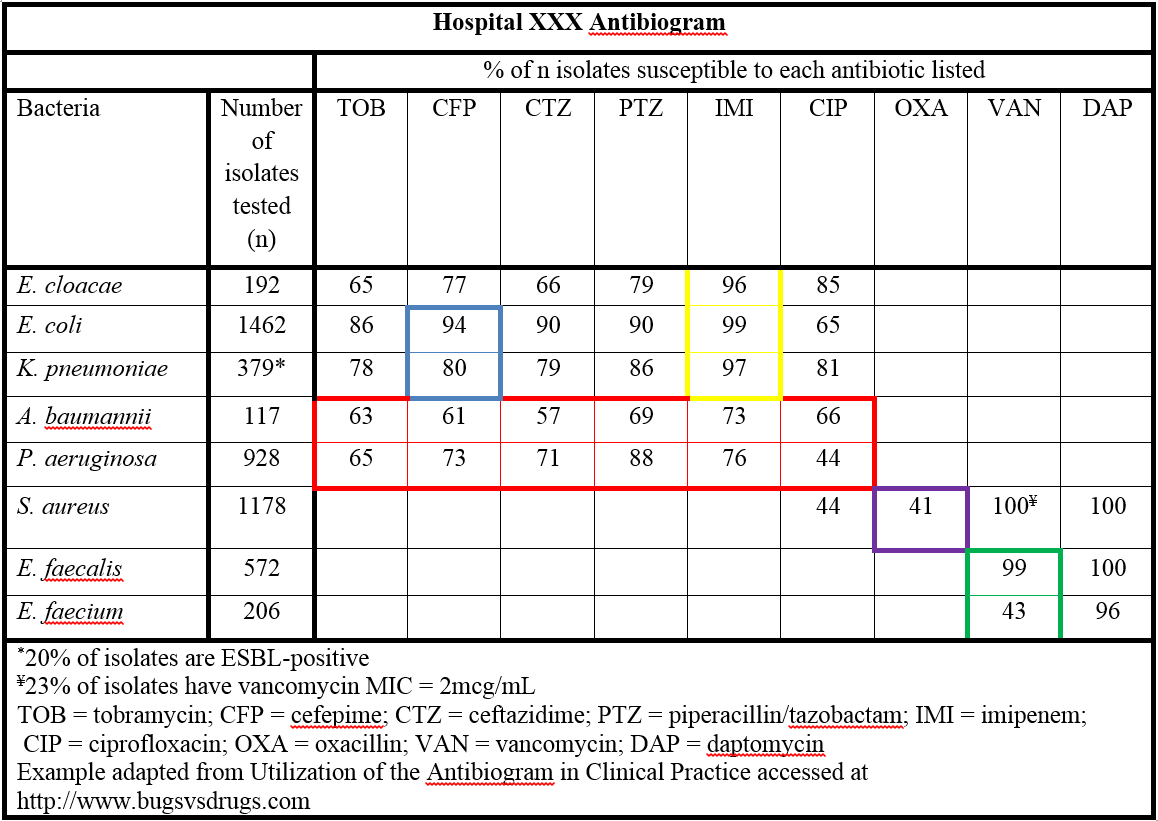
The antibiogram may be overwhelming to interpret. If broken down into sections, it is manageable. Figure 6, demonstrates how to read, interpret, and derive information about specific organisms of concern from an antibiogram.
Figure 6. Example hospital antibiogram.
Sections of the Antibiogram using the Example in Figure 6
Far left column: lists the names of the bacteria isolated in the laboratory and tested for antimicrobial sensitivity.
Second column from left: provides the number (n) of isolates reported for that particular genus and species. Another interpretation of “n” is number of patients identified with a potential infection caused by that pathogen. The frequency of isolates (n) represents only the first analyzed isolate per patient per CLSI guidelines. By not duplicating isolates from the same patient, clinicians will recognize which pathogens are the most common causes of infection. In the example in Figure 6, E. coli, S. aureus, and Pseudomonas species are the most common organisms at this institution.
Remaining columns from left to right: show the susceptibility rates (in percentages) to each of the different antimicrobials tested.
Interpretation of the Antibiogram through Scenarios and Example in Figure 6
Scenario #1. Treatment for infections with Pseudomonas aeruginosa or Acinetobacter baumannii.
When considering treatment for P. aeruginosa or A. baumannii, no single agent will exhibit excellent activity against either organism. In Figure 6, and outlined in red, all agents show limited susceptibility for these bacteria. Piperacillin/tazobactam (PTZ) looks superior though still limited. It is recommended to use combination therapy to ensure at least one antimicrobial is effective. Avoid two agents from the same class of antibiotics as there may be cross resistance. Using Figure 5 as an example, it is beneficial to avoid using both piperacillin/tazobactam and imipenem together. PTZ and ciprofloxacin or PTZ and tobramycin would be better combinations.
Scenario #2. Treatment of infections with Gram-negative pathogens.
When considering Gram-negative pathogens, an IP should know the prevalence of extended spectrum beta lactamase (ESBL) producing bacteria in their institution. These organisms are less susceptible to all beta lactam antibiotics, with the exception of the carbapenems. The frequency of ESBL producing organisms can be determined by looking at the cefepime (CFP) susceptibility. The blue box marked in Figure 6 describes 80% of K. pneumoniae isolates are susceptible to cefepime, which suggests that 20% likely produce the ESBL. Likewise, 94% of E. coli are susceptible to CFP, which suggests 6% are resistant and likely produce ESBL.
Scenario #3. Treatment of infections with organisms in the Enterobacteriaceae family.
Enterobacteriaceae are a family of Gram-negative bacteria that include both normal and pathogenic enteric microorganisms. Examples include species of Escherichia, Klebsiella, Enterobacter, Proteus, Providencia, and Serratia. Carbapenem resistant Enterobacteriaceae (CRE) include: Escherichia coli and Klebsiella species among others. Unlike resistance in methicillin-resistant Staphylococcus aureus (MRSA), carbapenem resistance is complex. It occurs in different Enterobacteriaceae and mediated by several mechanisms, including production of enzymes that inactivate carbapenems (carbapenemases). Two common enzymes are Klebsiella pneumoniae carbapenemase (KPC) and New Delhi metallo beta lactamase (NDM). Infections with CRE are difficult to treat. They resist most antibiotics, including the carbapenems, and are associated with mortality rates up to 50%. 13-15 Due to movement of patients in the healthcare system, CRE will be a problem in multiple facilities. CRE pose a serious threat to public health. The Centers for Disease Control and Prevention (CDC) has released, and continues to update, a CRE toolkit which expands on the 2009 CDC recommendations (see www.cdc.gov/hai/pdfs/cre/CRE-guidance-508.pdf). The box outlined in yellow in Figure 6 shows the percentage of members of the Enterobacteriaceae family (E. cloacae, E. coli and K. pneumoniae) susceptible to the one carbapenem antibiotic tested, imipenem. This suggests there may be some isolates that are CRE within this facility. Further testing is needed on those non-susceptible isolates to confirm the exact mechanism of resistance.
Confirmation testing in the laboratory, such as the Modified Hodge Test, can determine which type of resistance mechanism an organism has. These tests can be difficult and time-consuming and are not generally done in-house. The antibiogram can show the percentage of possible carbapenem-resistant Enterobacteriaceae to gain an approximation of potential CRE producing bacteria. This is done by looking at the Enterobacteriaceae sensitivity in the carbapenem column. The most utilized carbapenems in antimicrobial sensitivity testing include imipenem, meropenem, and ertapenem. The agent used to test for carbapenem sensitivity is institution specific.
Scenario #4. Treatment of MRSA and other Gram-positive infections.
When determining rates of methicillin-resistant Staphylococcus aureus (MRSA) it is important to look at susceptibility to oxacillin. Institutions may report nafcillin instead of oxacillin. Either one is a surrogate marker for methicillin and can be used interchangeably. The purple box in Figure 6 identifies 41% of S. aureus are susceptible to oxacillin, and therefore 59% are oxacillin resistant. Another interpretation would be 59% of S. aureus isolates are methicillin-resistant. No other beta-lactam has activity against MRSA except for the new cephalosporin, ceftaroline. Institutions should not report any other beta-lactam susceptibilities as different from oxacillin.
Scenario #5. Treatment of general infections caused by Staphylococcus aureus.
When evaluating S. aureus it is important to check the susceptibility to vancomycin. Generally vancomycin resistance is rare. It is helpful to know the distribution of MIC in the institution. S. aureus with a MIC of 2mcg/mL is considered susceptible by CLSI standards. There have been some clinical studies that show outcomes from S. aureus infections are suboptimal when treated with vancomycin when typical isolates in the institution had an MIC of 2mcg/mL.16, 17 While results of these studies are considered controversial, awareness of the situation is essential. If a clinician chooses to use vancomycin in this instance, a higher dose and careful monitoring of the patient are necessary to confirm appropriate responses. Figure 6 footnotes indicate, 23% of isolates have an MIC of 2mcg/mL. MIC values of the isolates should be reported as part of the antibiogram.
Scenario #6. Treatment of infections caused by Enterococci sp.
Species of Enterococci must be separated when reported on the antibiogram because Enterococcus faecium is predominantly vancomycin resistant.18, 19 As seen in the green box in Figure 6, the percent of Enterococci susceptible to vancomycin would be different if the two species were not separated and could result in prescribing an inappropriate empiric antibiotic.
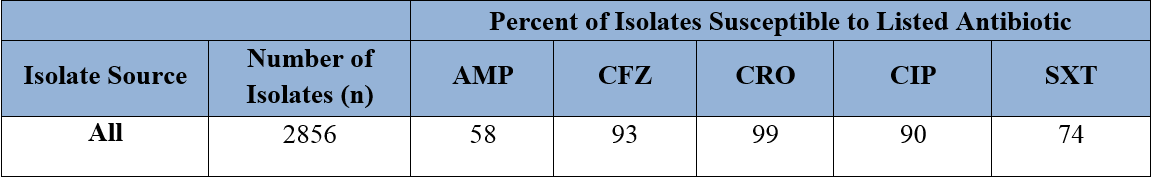
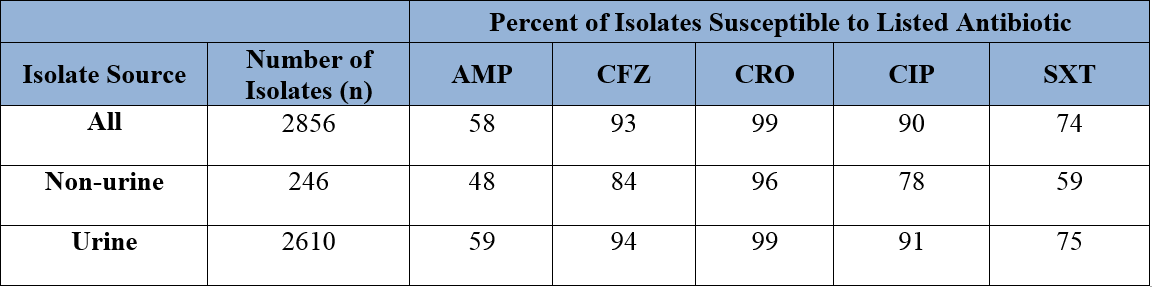
Scenario #7. Treatment of urinary tract infections and infections caused by Eserichia coli.
The antibiogram in Figure 6 includes urine isolates and explains why the number of E. coli isolates is large. The separation of urine vs. other sources provides a more accurate picture of non-urine E. coli susceptibility. Tables 2 and 3 provide an example of susceptibility data on an antibiogram when urine isolates are combined with (Table 2) or separated from (Table 3) other specimen sources. When tables 2 and 3 are compared, the susceptibility pattern differs greatly.
Table 2. All isolates of an organism grouped together. Abbreviations include: ampicillin (AMP); cefazolin (CFZ), ceftriaxone (CRO); ciprofloxacin (CIP); trimethoprim-sulfamethoxazole (SXT).
When interpreting the antibiogram, generalizations can be made:
- High MRSA rates usually mean poor infection practices20
- High VRE rates may be reduced by a decreased use of cephalosporins, particularly the third-generation cephalosporins such as ceftriaxone, ceftazidime, and cefotaxime21
- High ESBL rates may also be reduced by less use of the third generation cephalosporins, as ESBLs are often linked to third generation cephalosporin overuse22, 23
- High KPC rates are typically the result of overuse of cephalosporins and carbepenems23
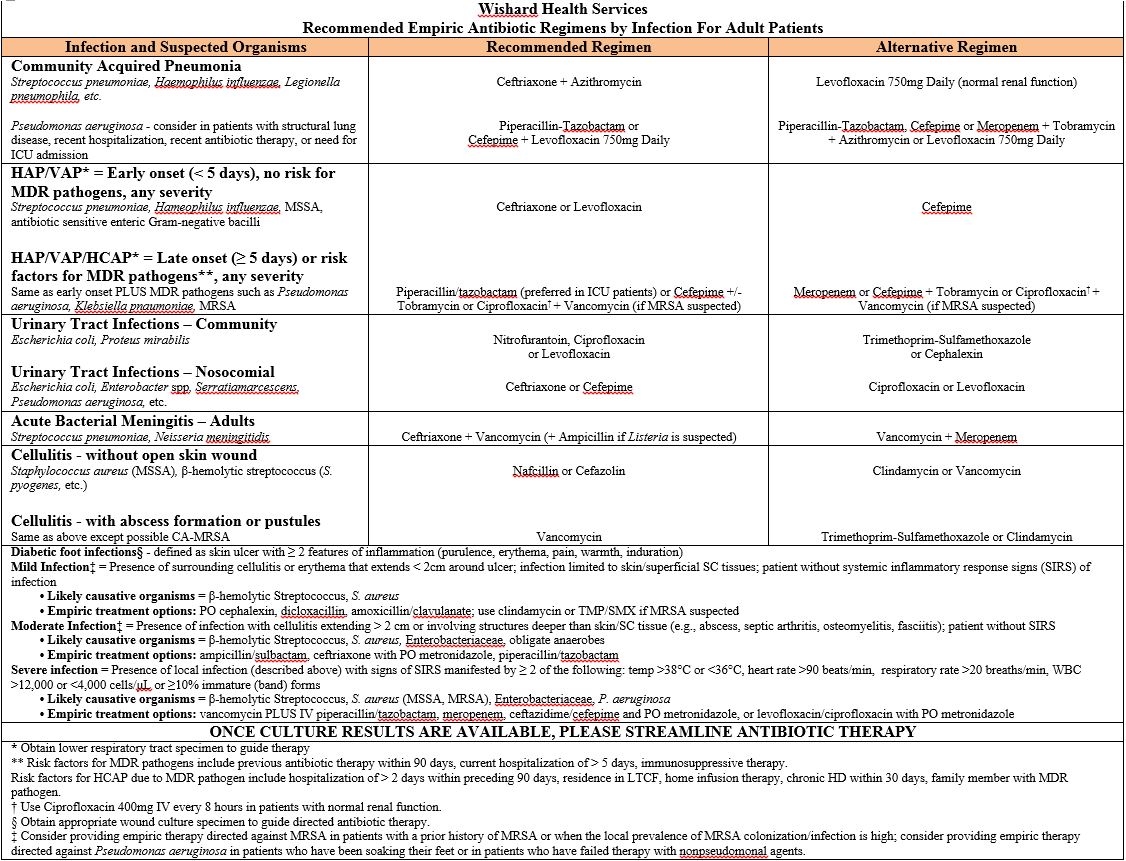
The antibiogram could be used to generate an “empiric antibiotic of choice” cheat sheet. This could be laminated and posted throughout the facility, made into pocket cards, and placed on the intranet along with the antibiogram. Figure 7 provides an example of a cheat sheet created from an antibiogram. Note the empiric antibiotic regimens shown in the example in Figure 7 are specific to that hospital. Remember that every facility must research its own susceptibility patterns. Antibiograms or empiric antibiotic regimens should not be used by facilities for which they were not originally created!!
Figure 7: Example empiric antibiotic regimens for a hospital. Used with permission from Sharon Erdman, Pharm D, Clinical Professor, Purdue University College of Pharmacy Infectious Diseases Clinical Pharmacist Co-Director Outpatient Parenteral Antimicrobial Therapy Program, Eskenazi Health.
Documentation and Reporting
Reporting/Distribution of the Antibiogram
Once the antibiogram has been prepared, distribute to those clinicians prescribing antibiotics and other persons needing easy access. The following list covers basic areas and/or people to distribute the antibiogram:
- Physicians
- Infection Preventionist
- Pharmacists
- Laboratory personnel
- On each hospital ward (e.g., emergency department, surgery, medical/surgical floor, intensive care units)
- Clinic rooms if the clinic utilizes the same laboratory
- Long term care center if it utilizes the same laboratory
- Express care if it utilizes the same laboratory
- Intranet website for the facility – Verify the antibiogram and related information can be quickly retrieved and is in an easy-to-use format. Educate personnel how to access the website.
- Antimicrobial stewardship committee. If the facility does not yet have an antimicrobial stewardship committee, then the infection prevention committee would review the antibiogram.
- Pharmacy and therapeutics (P&T) committee (as applicable). The P&T committee addresses matters pertaining to the use of medications within the institution, including pharmacy protocols and medication formulary review.
The antibiogram can be formatted in a variety of ways such as:
- Pocket cards
- Online documents
- Tabulated in a 3-ring binder
- Laminated in order to attach to walls
There are numerous layouts available, depending on where or how the antibiogram will be used. Use a cover page listing the facility, report date, and contact information. Site-specific information should include which area the isolates were taken from such as inpatient, outpatient (ED), medical wards, and intensive care units and whether or not the isolates were separated by specimen sources such as blood or urine. Items to consider including in the antibiogram are: disease state (e.g., pneumonia or urinary tract infection), patient demographics (e.g., age), or multi-drug resistant isolates (e.g., ESBLs, MRSA, VRE, or KPCs). When listing the antibiotics tested against the organism, consider including the breakpoints or MIC for susceptibility set by CLSI, particularly for vancomycin.
Reporting/Distribution of the Culture and Sensitivity Report
Who will be reviewing the culture and sensitivity (C&S) report will vary and will usually encompass more than one person. Key persons reviewing the C&S report include:
- Infection Preventionist (IP)
If possible the IP should review the C&S report on every patient as it becomes available. The IP should work with the microbiologist to develop a routing system for these reports to ensure appropriate tracking and timeliness. Options for routing culture and sensitivity reports between the laboratory and IP include:
- the IP receives an automated faxed report from the laboratory
- the report is sent to a folder on the intranet by the laboratory
- the IP physically retrieves a copy of the report from the laboratory on a daily basis
- It is best if a process can be in place to receive reports as they become available. Each facility will determine what works best given institution-specific time and cost constraints. Work with information technology personnel to assist with the automation of this report.
- Pharmacist – The pharmacist should review the C&S reports daily to ensure that they are correct and the optimal antibiotic is ordered. The pharmacist can recommend deescalating to a less broad spectrum antibiotic as soon as possible. This leads to less resistance development as well as potential cost savings. The pharmacist and the laboratory will need to coordinate the best approach for the transfer of information.
- Physician – The physician reviews the C&S report as soon as possible to ensure appropriate antibiotic use. Most often physicians will review the information in the electronic chart. If automation is not available, there must be a method for their quick notification. Options for efficient notification include: faxing the report to the physician’s office, calling the physician, or delivering the report in person. The process must be convenient and easy to use to ensure the optimal treatment for the patient.
- Microbiologist – The microbiologist reviews the report when it first becomes available. They should be trained to monitor and review potential trends, resistance patterns, and out-of-the-ordinary organisms, as well as contaminants that may need to be reported immediately. Procedures should be in place for reporting urgent situations, such as positive Clostridium difficile, Enterococcus species resistant to vancomycin, and Klebsiella pneumoniae resistant to imipenem (antibiotic) or other multidrug-resistant organisms.
- Nurse – Nurses play a role in reviewing the C&S reports as they continually utilize the patient’s chart. When a report is generated, the nurse should be able to interpret the results and compare the patient’s antibiotic regimen to the report. If the infection is noted as resistant to the antibiotic chosen for treatment, the nurse should immediately notify the physician.
- Antimicrobial Stewardship Program Committee (ASP) – The ASP should be notified of trends that may occur.
Common Issues Encountered
When instituting an antimicrobial stewardship program, common problems may be encountered. Table 4 describes common issues and potential solutions.
Table 4. Common issues encounters when implementing an antimicrobial stewardship program and possible solutions.
- Determine if the inclusion of each specific species is essential. If yes, it would be beneficial to also include a footnote in the antibiogram such as, “Organisms with n < 30 may not have statistically relevant susceptibility results. Interpret with caution.”
- The facility could consider combining multiple years of data such as every two years.
- Work together with other nearby facilities to generate a community antibiogram.
- Look for individuals in the facility routinely making suggestions on improving the antimicrobial care of the patient. Often this will be the IP and an interested pharmacist or microbiologist.
- Find any interested physician to be a champion. A potential candidate is the physician representing the infection prevention committee.
- Employ the specific physician to whom the IP reports.
Resources
Helpful/Related Readings
- Policy Statement on Antimicrobial Stewardship by the Society for Healthcare Epidemiology of America (SHEA), the Infectious Diseases Society of America (IDSA), and the Pediatric Infectious Diseases Society (PIDS). Available at: jstor.org/stable/10.1086/665010
- Analysis and Presentation of Cumulative Antibiograms: A New Consensus Guideline from the Clinical and Laboratory Standards Institute. Available at: oxfordjournals.org/content/44/6/867.full.pdf+html
- Dellit T., et al. Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America Guidelines for Developing an Institutional Program to Enhance Antimicrobial Stewardship. Clin Infect Dis 2007; 44:159-77. Available at: idsociety.org/uploadedFiles/IDSA/Guidelines-Patient_Care/PDF_Library/Antimicrobial%20Stewardship.pdf
- Grota P, Allen V, Boston KM, et al, eds. APIC Text of Infection Control & Epidemiolo 4th Edition. Washington, D.C.: Association for Professionals in Infection Control and Epidemiology, Inc.; 2014.
- Chapter 26, Antimicrobials and Resistance, by FW Arnold
- Chapter 110, Pharmacy Services, by ES Kastango, PC Kienle and K St. John
- Bennett J and Brachman P, eds. Bennett & Brachman’s Hospital Infection 6th Edition. Philadelphia, PA: William R Jarvis; 2014.
- Chapters14, Antimicrobial Stewardship: Programmatic Efforts to Optimize Antimicrobial Use, by RC Owens, Jr and WR Jarvis
- Chapter 15, Multidrug-Resistant Organisms: Epidemiology and Control, by MY Lin, RA Weinstein an MK Hayden
- Chapter 16, Molecular Biology of Resistance: A Brief History of Resistance mechanisms and the Discovery of Gene Transfer, by G Patel and RA Bonomo
- Chapter 17, Economic Evaluation of Healthcare-Associated Infections and Infection-Control and Antimicrobial-Stewardship Interventions, by EN Perencevich and SE Cosgrove
- Chapter 41, The Importance of Infection Control in Controlling Antimicrobial-Resistant Organisms, by CD Salgado and BM Farr
- Bennett G, Morrell G, and Green L, ed. Infection Prevention Manual for Hospitals; revised edition. Rome, GA: ICP Associates, Inc.; 2010. Section 7: pages 16-20
- Bennett G. Infection Prevention Manual for Ambulatory Care. Rome, GA: ICP Associates Inc.; 2009. Section 7: pages 16-20
- Bennett G and Kassai M. Infection Prevention Manual for Ambulatory Surgery Centers. Rome, GA: ICP Associates, Inc.; 2011. Section 7: pages 20-22
- The Sanford Guide To Antimicrobial Therapy (most current edition); available yearly in a wide array of formats. For more information or to purchase: sanfordguide.com
- Clinical and Laboratory Standards Institute (CLSI) M39: Guidelines For Reporting A Cumulative Antibiogram (current edition)
- Clinical and Laboratory Standards Institute (CLSI) M100-S23 Performance Standards for Antimicrobial Susceptibility Testing; Twenty-Third Informational Supplement
- Lautenbach E, Woeltje KF, and Malani PN, eds. SHEA Practical Healthcare Epidemiology (3rd Edition). University of Chicago Press, Chicago, IL; 2010
- Chapter 17 Control of Gram-Positive Multidrug-Resistant Pathogens, by T Van Schooneveld and ME Rupp
- Chapter 18 Control of Antibiotic-Resistant Gram-Negative Pathogens, by A Harris and K Thom
- Chapter 20 Improving Use of Antimicrobial Agents, by RA Duncan and KR Lawrence
- Mayhall CG, ed. Hospital Epidemiology and Infection Control (4th Edition). Philadelphia, PA: Lippincott Williams & Wilkins, a Wolters Kluwer business; 2011.
- Chapter 85, Mechanisms of Bacterial Resistance to Antimicrobial Agents, by U Stiefel and LB Rice
- Chapter 86, Antimicrobial Resistance and Healthcare-Associated Infection, by GL French
- Chapter 87, Antimicrobial Stewardship, by D Nathwani
Related Websites/Organizations
- Wyoming Department of Health, Infectious Disease Epidemiology Unit, Healthcare-Associated Infection Prevention; health.wyo.gov/phsd/epiid/HAIgeneral.html
- Mountain-Pacific Quality Health – Wyoming; mpqhf.com/wyoming/index.php
- American Society of Health-System Pharmacists; ashp.org
- Infectious Disease Society of America; idsociety.org
- Centers for Disease Control and Prevention; cdc.gov
- Utilization of the Antibiogram in Clinical Practice; bugsvsdrugs.com
- Clinical and Laboratory Standards Institute (CLSI); clsi.org/
Additional Resources
- Antibiogram Surveillance Method Using Cumulative Susceptibility Data. Accessed 12/22/13 cdc.gov/abcs/reports-findings/downloads/antibiogram-method.pdf
- Utilization of the Antibiogram in Clinical Practice. Accessed 12/22/13 bugsvsdrugs.com
- Images of antibiograms. Accessed 12/22/13 lexic.us/definition-of/antibiogram
- Hindler JF, Stelling J. Analysis and Presentation of Cumulative Antibiograms: A New Consensus Guideline from the Clinical and Laboratory Standards Institute. Accessed 12/20/13 cid.oxfordjournals.org/content/44/6/867.full.pdf+html
- Stanford Antibiogram errolozdalga.com/medicine/pages/OtherPages/stanfordAntibiogram.html
- CDC Get Smart for Healthcare Antibiotic Stewardship Measurement Framework. Accessed 12/21/13 cdc.gov/getsmart/healthcare/improve-efforts/driver-diagram/measurement-framework.html
- CDC Get Smart for Healthcare Keys for Success and Getting Started. Accessed 12/21/13 cdc.gov/getsmart/healthcare/improve-efforts/keys.html
- CDC Get Smart for Healthcare Antibiotic Stewardship Driver Diagram and Change Package. Accessed 12/21/13 cdc.gov/getsmart/healthcare/improve-efforts/driver-diagram/primary-driver1.html
- CDC Get Smart for Healthcare: Why Antimicrobial Stewardship? Accessed 12/21/13 cdc.gov/getsmart/healthcare/inpatient-stewardship.html
- CDC Get smart for Healthcare 2012 CRE Toolkit – Guidance for Control of Carbapenem-resistant Enterobacteriaceae (CRE). Accessed 12/22/13 cdc.gov/hai/organisms/cre/cre-toolkit/index.html
- Gilbert D., et al. The Sanford Guide To Antimicrobial Therapy 2013; 43:71-77
- Erdman S. Understanding the Hospital Antibiogram Webinar Accessed on 12/17/13
- Global RPh globalrph.com/
Helpful Contacts (in WY or US)
- Janet Hindler, Sr. Specialist, Clinical Microbiology at UCLA Medical Center, Consultant at Association of Public Health Laboratories, jhindler@ucla.edu
- Russ Forney, PhD, MT (ASCP), Surveyor, Wyoming Department of Health, 307-777-7123, forney@wyo.gov
- Mountain Pacific Quality Health (MPQH), (877) 810-6248
- Sharon Erdman, Pharm D, Clinical Professor, Purdue University College of Pharmacy Infectious Diseases Clinical Pharmacist Co-Director Outpatient Parenteral Antimicrobial Therapy Program, Eskenazi Health, 317-880-5423, serdman@iupui.edu
- Karen Burk RPh, Clinical Pharmacy Coordinator, Powell Valley Healthcare, Powell, WY 307-754-1179, kburk@pvhc.org
My Facility/City/County Contacts in this Area
Create a list of contacts that includes Name, phone number, and email, for people in the following positions:
- Lab director
- Pharmacy Director
- Infectious Disease Physician
- Antimicrobial Stewardship Committee Contacts
- Infection Preventionist
- Microbiologist
- Physician
- Pharmacist
- RN – Med./Surg.
- RN – LTCC
- RN-Clinic
- RN – ICU
- Others
- Pharmacy & Therapeutics Committee Chairperson
- Surveyor, WY Dept. of Health
- County of Local Public Health Contact
- Mountain-Pacific Quality Health – Wyoming Contact
References
- Clinical and Laboratory Standards Institute (CLSI) M39-A: Guidelines For Reporting A Cumulative Antibiogram Current Edition. M39-A Vol. 22(8):7
- Patel G, Huprikar S, Factor SH, Jenkins SG, Calfee DP. Outcomes of carbapenem-resistant Klebsiella pneumoniae infection and the impact of antimicrobial and adjunctive therapies. Infect Control Hosp Epidemiol 2008 Dec;29(12):1099–106
- Schwaber MJ, Lev B, Israeli A, et al. Containment of a country-wide outbreak of carbapenem-resistant Klebsiella pneumoniae in Israeli hospitals via a nationally implemented intervention. Clin Infect Dis 2011;52:848–55
- Jacob JT, Klein E, et al. Vital Signs: Carbapenem-Resistant Enterobacteriaceae. Morbidity and Mortality Weekly Report (MMWR) March 8, 2013 / 62(09);165-170 cdc.gov/mmwr/preview/mmwrhtml/mm6209a3.htm Accessed 12/21/2013
- Rybak M, Lomaestro B,et al.Therapeutic monitoring of vancomycin in adult patients: A consensus review of the American Society of Health-System Pharmacists, the Infectious Diseases Society of America, and the Society of Infectious Diseases Pharmacists. Am J Health-Syst Pharm. 2009; 66:82-98
- Lodise TP, Graves J, et al. Relationship between Vancomycin MIC and Failure among Patients with Methicillin-Resistant Staphylococcus aureus Bacteremia Treated with Vancomycin. Agents Chemother. September 2008; 52:9 3315-3320
- Mutters, NT, Mersch-Sundermann, V. et al. Control of the Spread of Vancomycin-Resistant Enterococci in Hospitals: Epidemiology and Clinical Relevance. Dtsch Arztebl Int 2013; 110(43): 725-31
- Cetinkaya, Y, Falk, P, Mayhall CG. Vancomycin-Resistant Enterococci. Clin Microbiol Rev. Oct 2000; 13(4): 686–707
- Allegranzi B, Pittet D The role of hand hygiene in healthcare-associated infection prevention. J Hosp Infect 2009;73: 305–315
- Fridkin SK, Edwards JR, et al. The Effect of Vancomycin and Third-Generation Cephalosporins on Prevalence of Vancomycin-Resistant Enterococci in 126 U.S. Adult Intensive Care Units. Annals of Internal Medicine 2001;135: 175–183
- Urbánek K, Kolár M, et al. Influence of Third-Generation Cephalosporin Utilization on the Occurrence of ESBL-positive Klebsiella pneumoniae Strains. J Clin Pharm Ther. 2007;13(4):403–8
- Meyer E, Schwab F, et al. Dramatic increase of third-generation cephalosporin-resistant E. coli in German intensive care units: secular trends in antibiotic drug use and bacterial resistance, 2001 to 2008. Crit Care. 2010;14(3):R113
- Dellit T., et al. Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America Guidelines for Developing an Institutional Program to Enhance Antimicrobial Stewardship. Clin Infect Dis 2007; 44:159-77; idsociety.org/uploadedFiles/IDSA/Guidelines-Patient_Care/PDF_Library/Antimicrobial%20Stewardship.pdf Accessed 12/22/2013
- Muto CA, Blank MK, et al. Control of an Outbreak of an Infection with the Hypervirulent Clostridium difficile BI strain in a University hospital using a comprehensive “bundle” approach. CID 2007, 45: 1266-1273
- Przybylski KG, Rybak MJ, Martin PR, et al. A pharmacist-initiated program of intravenous to oral antibiotic conversion. Pharmacotherapy 1997 Mar-Apr;17(2):271-6
- Bond, Cynthia L. Raehl. Clinical and Economic Outcomes of Pharmacist Managed Aminoglycoside or Vancomycin Therapy C.A. (CAB). Am J Health Syst Pharm. 2005;62(15):1596-1605
- Camins BC, King MD, Wells JB, et al. Impact of an antimicrobial utilization program on antimicrobial use at a large teaching hospital: a randomized controlled trial. Infection Control and Hospital Epidemiology Oct 2009;30(10):931-938
- Patel SJ, Saiman L, et al. Development of an antimicrobial stewardship intervention using a model of actionable feedback. Interdiscip Perspect Infect Dis. 2012; 2012: 150367
- Gauthier TP, Lantz E, et al. Internet-Based Institutional Antimicrobial Stewardship Program Resources in Leading US Academic Medical Centers. Clin Infect Dis. Feb 2014;58(3):445-446
- Society of Infectious Diseases Pharmacists. Antimicrobial Stewardship: A Certificate Program for Pharmacists. 2012; sidp.org/Default.aspx?pageId=1442823 Accessed 2/24/2014
- Centers for Disease Control and Prevention (CDC). Implementing and Improving Stewardship Efforts. cdc.gov/getsmart/healthcare/improve-efforts/index.html Accessed 1/4/14
- Leekha S, Terrell CL, Edson RS. General principles of antimicrobial therapy. Mayo Clin Proc. Feb 2011;86(2): 156–167 ncbi.nlm.nih.gov/pmc/articles/PMC3031442/
- Mueller M1, de la Peña A, Derendorf H Issues in pharmacokinetics and pharmacodynamics of anti-infective agents: kill curves versus MIC. Antimicrob Agents Chemother. 2004 Feb;48(2):369-77 ncbi.nlm.nih.gov/pmc/articles/PMC321563/
Appendices
Please download the PDF Version of this section, linked at the top of this page, to view the following appendices:
Appendix A: CDC’s Antibiotic Stewardship Driver Program
Appendix B: CDC’s Checklist for Core Elements of Hospital Antibiotic Stewardship Programs
Appendix C: List of Bacterial Organisms and Appropriate Antimicrobials (and associated exercise)

WIPAG welcomes your comments and feedback on these sections.
For comments or inquiries, please contact:
Cody Loveland, MPH, Healthcare-Associated Infection (HAI) Prevention Coordinator
Infectious Disease Epidemiology Unit,
Public Health Sciences Section, Public Health Division
Wyoming Department of Health
6101 Yellowstone Road, Suite #510
Cheyenne, WY 82002
Tel: 307-777-8634 Fax: 307-777-5573
Email: cody.loveland@wyo.gov